Introduction:
Guanidine carbonate, also referred to as carbamic guanidine, is a significant chemical compound widely utilized in diverse industries. Chemically represented by the formula C5H14N2O3, guanidine carbonate is formed through the reaction between hexamethylenediamine and carbonic acid. Its molecular structure, H2N(CH2)4NHCO2H, underscores its importance as a versatile intermediate in chemical synthesis and manufacturing processes.
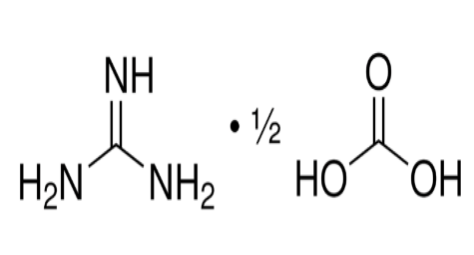
Product Features:
Guanidine carbonate typically presents as a white crystalline solid, soluble in water and common organic solvents. It possesses moderate alkalinity and exhibits reactivity as both a base and a nucleophile. With amino (-NH2) and carboxyl (-COOH) functional groups, guanidine carbonate offers versatility in chemical reactions, enabling the synthesis of a wide range of organic compounds.

Applications:
Pharmaceutical Synthesis: Guanidine carbonate serves as a key intermediate in pharmaceutical synthesis, contributing to the production of various medications, including antiviral drugs, antidiabetic agents, and muscle relaxants.
Polymer Industry: Guanidine carbonate finds application in the polymer industry as a crosslinking agent and catalyst in the synthesis of polyurethanes, resins, and specialty polymers.
Cosmetics: In the cosmetics industry, guanidine carbonate is used in hair straightening products and as a pH adjuster in skincare formulations.

Photography: Guanidine carbonate is employed in the production of photographic chemicals, particularly as a stabilizer in photographic developers.

Conclusion:
Guanidine carbonate emerges as a versatile and indispensable chemical compound in various industrial sectors. Its role as a key intermediate in pharmaceuticals, polymers, cosmetics, and photography underscores its significance in chemical synthesis and manufacturing processes.
View More Articals via www.fyyschemistry.com.
