Introduction:
Hydrochloric guanidine, with a chemical formula of HCl·C5H14N2, represents a pivotal chemical compound renowned for its diverse applications across various industries. Its molecular structure expressed as H2N(CH2)4NH2·HCl, emerges from the reaction between hexamethylenediamine and hydrochloric acid. Widely utilized in both chemical engineering and laboratory settings, hydrochloric guanidine stands out for its multifunctionality and stability.
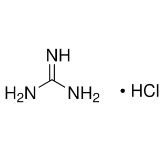
Product Features:
Hydrochloric guanidine exists as a white crystalline solid, readily soluble in water at room temperature, showcasing considerable reducing and alkaline properties. The presence of two amino (-NH2) groups and one chloride ion in its molecular structure endows it with excellent chemical reactivity. While stable in dry air, precautions should be taken to avoid contact with oxidizing agents to prevent undesired reactions.

Applications:
Metal Surface Treatment: Hydrochloric guanidine serves as a valuable metal surface treatment agent, facilitating the cleaning and removal of oxide layers and impurities from metal surfaces, and preparing them for subsequent coating or processing.
Organic Synthesis: Hydrochloric guanidine plays a pivotal role in chemical transformations as a key intermediate in organic synthesis. It finds utility in the synthesis of various organic compounds, including polymers and pharmaceutical intermediates.
Water Treatment: Employed as a water treatment agent, hydrochloric guanidine aids in pH adjustment, removal of metal ions, and impurities from water, and serves as a buffering agent to stabilize water quality.

Pharmaceutical Industry: In the pharmaceutical industry, hydrochloric guanidine is utilized as a pharmaceutical intermediate for synthesizing various medications, including anticancer agents and antidepressants.
Hydrochloric guanidine’s multifunctionality and reliability render it indispensable across various sectors, making it a preferred choice in many industrial and laboratory applications.
View More Articals via www.fyyschemistry.com.
